Overview:
7 Minutes
In this video, Craig Lipset, founder and advisor of Clinical Innovation Partners and co-chair of the Decentralized Trials and Research Alliance (DITRA), discusses the critical shortage of Clinical Research Associates (CRAs) in the clinical trial industry.
Topics discussed:
- The need for adaptability and willingness to take different approaches
- Not relying on past assumptions and expectations to solve the problem
- How the industry has rapidly evolved and adopted new approaches during the pandemic
- The impact of new trial models and technology on the role of CRAs
- The importance of creating a more diverse clinical workforce and how it can help in recruiting clinical trial subjects
More about the speaker:
Craig Lipset (he/him/his) leads at the forefront of innovation in clinical research and medicine development. He is an advisor to technology and biopharmaceutical companies, leading universities, and the venture community, bringing vision and driving action at the intersection of research, digital solutions, and patient engagement. Craig was the Head of Clinical Innovation and Venture Partner at Pfizer, on the founding Operations Committee for TransCelerate Biopharma, and on the founding management teams for two successful startup ventures (Perceptive Informatics and Adnexus Therapeutics). During that time, Craig designed and launched multiple industry firsts — from the first fully remote/virtual clinical trial for a new medicine to the first returning of results and data to research participants.
He currently serves on the Board of Directors for the Decentralized Trials & Research Alliance (Co-Chair), Foundation for Sarcoidosis Research and the MedStar Health Research Institute, and, as well as on the Editorial Board for Therapeutic Innovation & Regulatory Science. Craig is an Adjunct Assistant Professor in Health Informatics at Rutgers University, and an Adjunct Instructor at the University of Rochester Center for Health + Technology.
Craig has received the red jacket ‘hall of fame’ recognition among the PharmaVOICE most inspiring people in the life sciences. He has been recognized among Pharmaceutical Executive’s Emerging Leaders, CenterWatch Top 20 Innovators in Clinical Trials, the Medicine Maker Power List and the AlleyWatch Who’s Who in eHealth. He studied Music at Brandeis University and earned a Master of Public Health from Columbia.
LinkedIn: linkedin.com/in/lipset/
Twitter: twitter.com/craiglipset
YouTube: http://bit.ly/CraigTalksClinical
Watch the video now, no registration is required
Other educational videos
Industry expert interviews
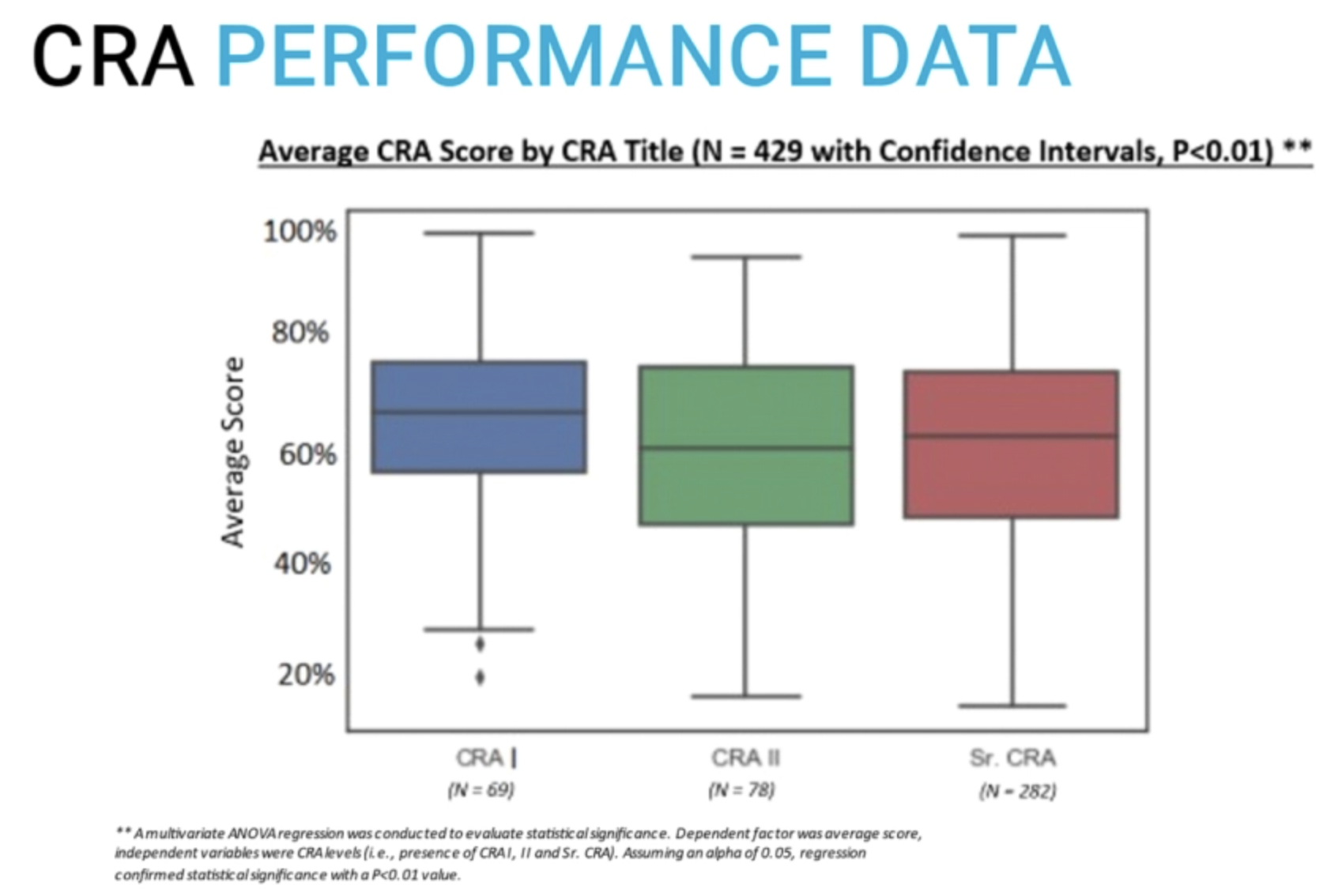
Experience did not have any association with performance 14:24
Moe Alsumidaie, Head of Research for CliniBiz, recently conducted a study on the correlation of CRA experience with performance.
Clinical trial sites are a critical part of the supply chain 10:14
How one organization challenged the standard for hiring clinical research talent by training new talent to meet the demands of the industry.
Strategies to address the shortage of clinical research talent. 7:11
Rhonda emphasizes the importance of mentoring and hands-on training for new employees, as not everyone comes into the industry with the same level of experience and capability.